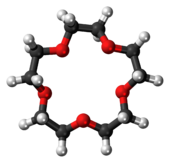
15-Crown-5

| |
| |
| Names | |
|---|---|
| Preferred IUPAC name
1,4,7,10,13-Pentaoxacyclopentadecane[1] | |
| Identifiers | |
3D model (JSmol)
|
|
| 1618144 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.046.694 |
| EC Number |
|
| 3897 | |
| MeSH | 15-Crown-5 |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C10H20O5 | |
| Molar mass | 220.265 g·mol−1 |
| Appearance | Clear, colorless liquid |
| Density | 1.113 g cm−3 (at 20 °C) |
| Boiling point | 93–96 °C (199–205 °F; 366–369 K) at 0.05 mmHg |
| log P | -0.639 |
Refractive index (nD)
|
1.465 |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
-881.1--877.1 kJ mol−1 |
Std enthalpy of
combustion (ΔcH⦵298) |
-5.9157--5.9129 MJ mol−1 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H315, H319 | |
| P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
| Flash point | 113 °C (235 °F; 386 K) |
| Safety data sheet (SDS) | msds.chem.ox.ac.uk |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
15-Crown-5 is a crown ether with the formula (C2H4O)5. It is a cyclic pentamer of ethylene oxide that forms complex with various cations, including sodium (Na+)[2] and potassium (K+);[3] however, it is complementary to Na+ and thus has a higher selectivity for Na+ ions.
Synthesis
[edit]15-Crown-5 can be synthesized using a modified Williamson ether synthesis:[4]
- (CH2OCH2CH2Cl)2 + O(CH2CH2OH)2 + 2 NaOH → (CH2CH2O)5 + 2 NaCl + 2 H2O
It also forms from the cyclic oligomerization of ethylene oxide in the presence of gaseous boron trifluoride.[5]
Properties
[edit]Analogous to 18-crown-6, 15-crown-5 binds to sodium ions. Thus, when treated with this complexing agent, sodium salts often become soluble in organic solvents.
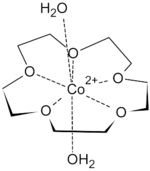
First-row transition metal dications fit snugly inside the cavity of 15-crown-5. They are too small to be included in 18-crown-6. The binding of transition metal cations results in multiple hydrogen-bonded interactions from both equatorial and axial aqua ligands, such that highly crystalline solid-state supramolecular polymers can be isolated. Metal salts isolated in this form include Co(ClO4)2, Ni(ClO4)2, Cu(ClO4)2, and Zn(ClO4)2. Seven coordinate species are most common for transition metal complexes of 15-crown-5, with the crown ether occupying the equatorial plane, along with 2 axial aqua ligands.[6]
15-crown-5 has also been used to isolate salts of oxonium ions. For example, from a solution of tetrachloroauric acid, the oxonium ion [H7O3]+ has been isolated as the salt [(H7O3)(15-crown-5)2][AuCl4]. Neutron diffraction studies revealed a sandwich structure, which shows a chain of water with remarkably long O-H bond (1.12 Å) in the acidic proton, but with a very short OH•••O distance (1.32 Å).[6]

A derivative of 15-crown-5, benzo-15-crown-5, has been used to produce anionic complexes of carbido ligands as their [K(benzo-15-crown-5)2]+ salts:[6]
- (Ar2N)3MoCH + KCH2Ph + 2 (15-crown-5) → [K(15-crown-5)2]+[(Ar2N)3MoC]− + CH3Ph
See also
[edit]References
[edit]- ^ "15-crown-5 - Compound Summary". PubChem Compound. USA: National Center for Biotechnology Information. 16 September 2004. Identification and Related Records. Retrieved 11 October 2011.
- ^ Takeda, Y.; et al. (1988). "A Conductance Study of 1:1 Complexes of 15-Crown-5, 16-Crown-5, and Benzo-15-crown-5 with Alkali Metal Ions in Nonaqueous Solvents". Bulletin of the Chemical Society of Japan. 61 (3): 627–632. doi:10.1246/bcsj.61.627.
- ^ Chen, Chun-Yen; et al. (2006). "Potassium ion recognition by 15-crown-5 functionalized CdSe/ZnS quantum dots in H2O". Chem. Comm. (3): 263–265. doi:10.1039/B512677K. PMID 16391728.
- ^ Cook, Fred L.; Caruso, Thomas C.; Byrne, Michael P.; Bowers, Chauncey W.; Speck, Don H.; Liotta, Charles L. (1974). "Facile syntheses of 12-crown-4 and 15-crown-5". Tetrahedron Letters. 15 (46): 4029–4032. doi:10.1016/S0040-4039(01)92075-1.
- ^ Liotta, Charles L.; Berkner, Joachim (2001), "15-Crown-5", Encyclopedia of Reagents for Organic Synthesis, Chichester, UK: John Wiley & Sons, Ltd, doi:10.1002/047084289x.rc263, ISBN 978-0-471-93623-7
- ^ a b c Jonathan W. Steed; Jerry L. Atwood (2009). Supramolecular Chemistry, 2nd edition. Wiley. ISBN 978-0-470-51233-3.
Further reading
[edit]- Klok, H.A.; et al. (1997). "Novel benzo-15-crown-5 functionalized α-olefin/CO terpolymers for membrane applications". Macromolecular Chemistry and Physics. 198 (9): 2759–2768. doi:10.1002/macp.1997.021980908.
- Fedorova, O.A.; et al. (2005). "Facile synthesis of novel styryl ligands containing a 15-crown-5 ether moiety". Arkivoc. xv: 12–24. doi:10.3998/ark.5550190.0006.f03. hdl:2027/spo.5550190.0006.f03.

