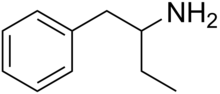
Phenylisobutylamine
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C10H15N |
| Molar mass | 149.237 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Phenylisobutylamine, also known as α-ethylphenethylamine, Butanphenamine, B or AEPEA,[1] is a stimulant drug of the phenethylamine class. It is a higher homologue of amphetamine, differing from amphetamine's molecular structure only by the substitution of the methyl group at the alpha position of the side chain with an ethyl group. Compared to amphetamine, phenylisobutylamine has strongly reduced dopaminergic effects, and instead acts as a selective norepinephrine releasing agent.[citation needed] The dextroisomer of phenylisobutylamine partially substitutes for dextroamphetamine in rats.[1]
A number of derivatives of phenylisobutylamine are known, including BDB, MBDB, EBDB, butylone (βk-MBDB), eutylone (βk-EBDB), Ariadne (α-Et-DOM), 4-CAB, and 4-MAB.
"Phenylisobutylamine" is in fact a chemical misnomer because isobutylamine itself contains a branched chain. The correct name after this style for this class of compound would be "phenylsecbutylamine".
See also
[edit]References
[edit]| Phenethylamines |
|
|---|---|
| Amphetamines |
|
| Phentermines |
|
| Cathinones |
|
| Phenylisobutylamines | |
| Phenylalkylpyrrolidines | |
| Catecholamines (and close relatives) |
|
| Miscellaneous |
|
- Articles with short description
- Short description is different from Wikidata
- Drugs with non-standard legal status
- Articles without EBI source
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- All articles with unsourced statements
- Articles with unsourced statements from April 2010
